Stratospheric Ozone Depletion
1. Where do we find the ozone layer?
(source: https://www.epa.gov/ozone-layer-protection/basic-ozone-layer-science)
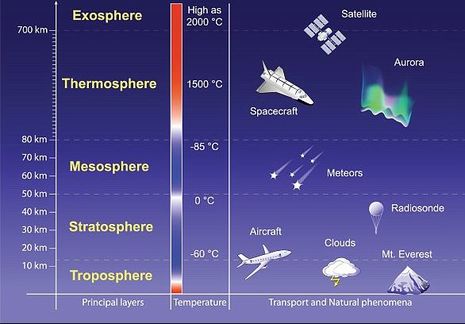
The Earth's atmosphere is composed of several layers. The lowest layer, the troposphere, extends from the Earth's surface up to about 6 miles or 10 kilometers (km) in altitude. Virtually all human activities occur in the troposphere. Mt. Everest, the tallest mountain on the planet, is only about 5.6 miles (9 km) high. The next layer, the stratosphere, continues from 6 miles (10 km) to about 31 miles (50 km). Most commercial airplanes fly in the lower part of the stratosphere.
Most atmospheric ozone is concentrated in a layer in the stratosphere, about 9 to 18 miles (15 to 30 km) above the Earth's surface (see the figure below). Ozone is a molecule that contains three oxygen atoms. At any given time, ozone molecules are constantly formed and destroyed in the stratosphere. The total amount has remained relatively stable during the decades that it has been measured.
The ozone layer in the stratosphere absorbs a portion of the radiation from the sun, preventing it from reaching the planet's surface. Most importantly, it absorbs the portion of UV light called UVB. UVB a band of ultraviolet radiation with wavelengths from 280-320 nanometers produced by the Sun. UVB is a kind of ultraviolet light from the sun (and sun lamps) that has several harmful effects, particularly effective at damaging DNA. It is a cause of melanoma and other types of skin cancer. It has also been linked to damage to some materials, crops, and marine organisms.
(source: https://www.epa.gov/ozone-layer-protection/basic-ozone-layer-science)
The Earth's atmosphere is composed of several layers. The lowest layer, the troposphere, extends from the Earth's surface up to about 6 miles or 10 kilometers (km) in altitude. Virtually all human activities occur in the troposphere. Mt. Everest, the tallest mountain on the planet, is only about 5.6 miles (9 km) high. The next layer, the stratosphere, continues from 6 miles (10 km) to about 31 miles (50 km). Most commercial airplanes fly in the lower part of the stratosphere.
Most atmospheric ozone is concentrated in a layer in the stratosphere, about 9 to 18 miles (15 to 30 km) above the Earth's surface (see the figure below). Ozone is a molecule that contains three oxygen atoms. At any given time, ozone molecules are constantly formed and destroyed in the stratosphere. The total amount has remained relatively stable during the decades that it has been measured.
The ozone layer in the stratosphere absorbs a portion of the radiation from the sun, preventing it from reaching the planet's surface. Most importantly, it absorbs the portion of UV light called UVB. UVB a band of ultraviolet radiation with wavelengths from 280-320 nanometers produced by the Sun. UVB is a kind of ultraviolet light from the sun (and sun lamps) that has several harmful effects, particularly effective at damaging DNA. It is a cause of melanoma and other types of skin cancer. It has also been linked to damage to some materials, crops, and marine organisms.
2. Ozone Depletion
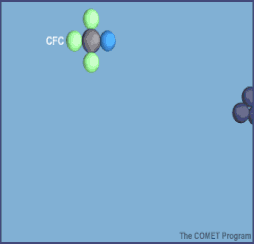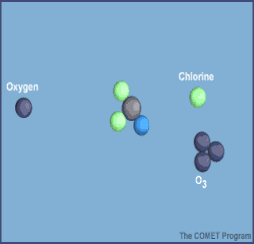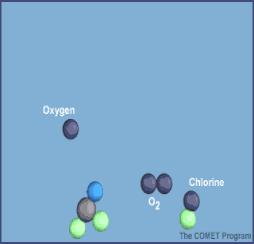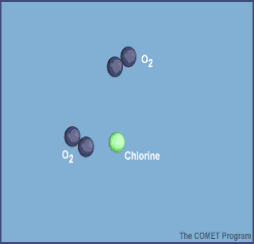
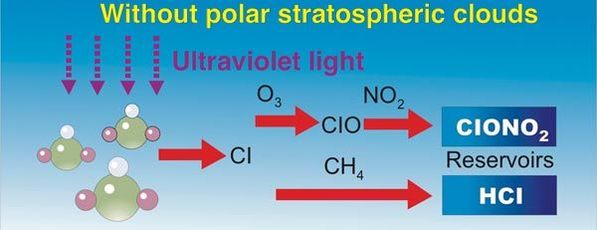
Here are diagrams of how the chlorine molecules react with the ozone molecules.
| ||||||
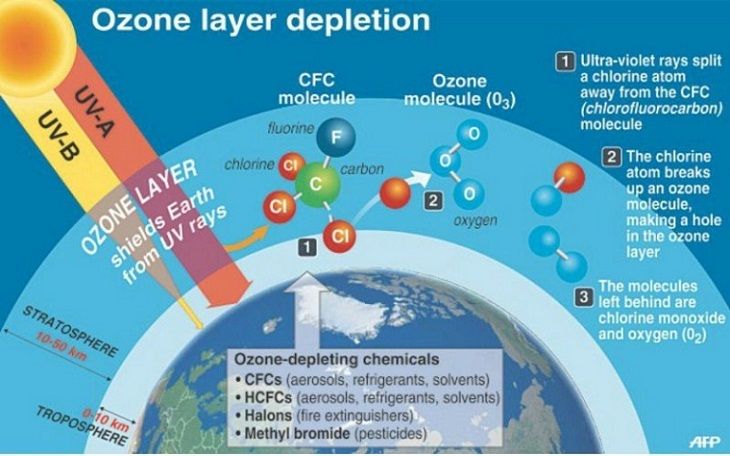
When chlorine and bromine atoms come into contact with ozone in the stratosphere, they destroy ozone molecules. One chlorine atom can destroy over 100,000 ozone molecules before it is removed from the stratosphere. Ozone can be destroyed more quickly than it is naturally created.
Some compounds release chlorine or bromine when they are exposed to intense UV light in the stratosphere. These compounds contribute to ozone depletion, and are called ozone-depleting substances (ODS). ODS that release chlorine include chlorofluorocarbons (CFCs), hydrochlorofluorocarbons (HCFCs), carbon tetrachloride, and methyl chloroform. ODS are generally very stable in the troposphere and only degrade under intense ultraviolet light in the stratosphere. When they break down, they release chlorine or bromine atoms, which then deplete ozone.
A detailed list of class I and class II substances with their ODPs, GWPs, and CAS numbers are available. (http://www.epa.gov/ozone/science/ods/index.html)
Gases covered under the 1987 Montreal Protocol and used for refrigeration, air conditioning, packaging, insulation, solvents, or aerosol propellants. Since they are not destroyed in the lower atmosphere, CFCs drift into the upper atmosphere where, given suitable conditions, they break down ozone. These gases are being replaced by other compounds: hydrochlorofluorocarbons, an interim replacement for CFCs that are also covered under the Montreal Protocol, and hydrofluorocarbons, which are covered under the Kyoto Protocol.
In the 1970s, concerns about the effects of ozone-depleting substances on the stratospheric ozone layer prompted several countries, including the United States, to ban the use of chlorofluorocarbons (CFCs) as aerosol propellants. However, global production of CFCs and other ODS continued to grow rapidly as new uses were found for these chemicals in refrigeration, fire suppression, foam insulation, and other applications.
Some natural processes, such as large volcanic eruptions, can have an indirect effect on ozone levels. For example, Mt. Pinatubo's 1991 eruption did not increase stratospheric chlorine concentrations, but it did produce large amounts of tiny particles called aerosols. These aerosols increase chlorine's effectiveness at destroying ozone. The aerosols in the stratosphere create a surface on which CFC-based chlorine can destroy ozone. However, the effect from volcanoes is short-lived.
Not all chlorine and bromine sources contribute to ozone layer depletion. For example, researchers have found that chlorine from swimming pools, industrial plants, sea salt, and volcanoes does not reach the stratosphere. In contrast, ODS are very stable and do not dissolve in rain. Thus, there are no natural processes that remove the ODS from the lower atmosphere.
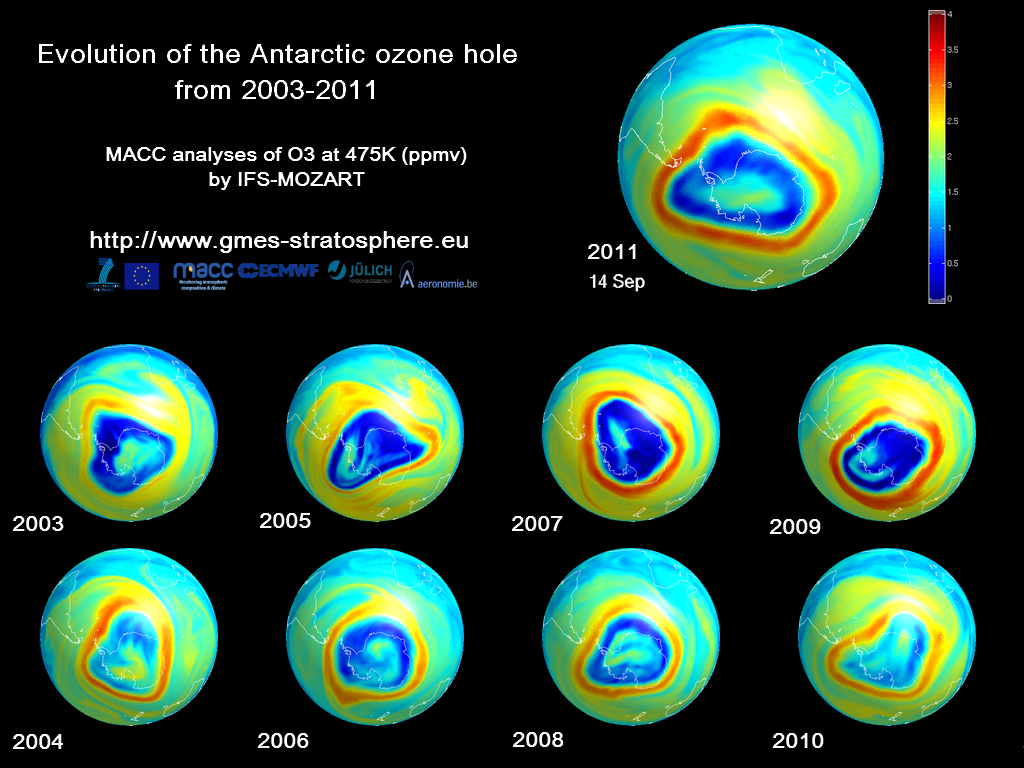
One example of ozone depletion is the annual ozone "hole" over Antarctica that has occurred during the Antarctic spring since the early 1980s. This is not really a hole through the ozone layer, but rather a large area of the stratosphere with extremely low amounts of ozone.
Ozone depletion is not limited to the area over the South Pole. Research has shown that ozone depletion occurs over the latitudes that include North America, Europe, Asia, and much of Africa, Australia, and South America. More information about the global extent of ozone depletion can be found in the Scientific Assessment of Ozone Depletion: 2014 developed by the United Nations Environment Programme.
|
|
|
3. The problems with ozone depletion?
As you go through the information below. Think of the following exam style questions.
- To what extent could stratospheric ozone depletion decrease the carrying capacity of an area?
- Discuss the impact of stratospheric ozone on human food systems.
|
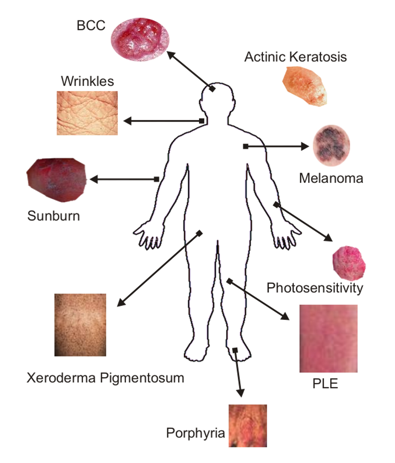
Human impact: Ozone layer depletion increases the amount of UVB that reaches the Earth’s surface. Laboratory and epidemiological studies demonstrate that UVB causes non-melanoma skin cancer and plays a major role in malignant melanoma development. In addition, UVB has been linked to the development of cataracts, a clouding of the eye’s lens. Because all sunlight contains some UVB, even with normal stratospheric ozone levels, it is always important to protect your skin and eyes from the sun. See a more detailed explanation of health effects linked to UVB exposure. Effects on plants UVB radiation affects the physiological and developmental processes of plants. Despite mechanisms to reduce or repair these effects and an ability to adapt to increased levels of UVB, plant growth can be directly affected by UVB radiation. Indirect changes caused by UVB, such as changes in plant form, how nutrients are distributed within the plant. Changes can have important implications for plant competitive balance, herbivory, plant diseases, and biogeochemical cycles. Effects on marine ecosystems Phytoplankton form the foundation of aquatic food webs. Phytoplankton productivity is limited to the euphotic zone, the upper layer of the water column in which there is sufficient sunlight to support net productivity. Exposure to solar UVB radiation has been shown to affect both orientation and motility in phytoplankton, resulting in reduced survival rates for these organisms. Scientists have demonstrated a direct reduction in phytoplankton production due to ozone depletion-related increases in UVB. UVB radiation has been found to cause damage to early developmental stages of fish, shrimp, crab, amphibians, and other marine animals. The most severe effects are decreased reproductive capacity and impaired larval development. Small increases in UVB exposure could result in population reductions for small marine organisms with implications for the whole marine food chain. Effects on materials: Synthetic polymers, naturally occurring biopolymers, as well as some other materials of commercial interest are adversely affected by UVB radiation. Today's materials are somewhat protected from UVB by special additives. Yet, increases in UVB levels will accelerate their breakdown, limiting the length of time for which they are useful outdoors. |
4. Ozone management
Through the 1970s and the 1980s, the international community became increasingly concerned that ODS would harm the ozone layer. In 1985, the Vienna Convention for the Protection of the Ozone layer formalised international cooperation on this issue. This cooperation resulted in the signing of the Montreal Protocol on Substances that Deplete the Ozone layer in 1987. After the Montreal Protocol was signed, new data showed worse-than-expected damage to the ozone layer. In 1992, the Parties to the Protocol decided to alter the terms of the 1987 agreement to end production of halons by 1994 and CFCs by 1996 in developed countries.
The original Montreal Protocol, signed in 1987, was the first step in international efforts to protect stratospheric ozone. Under the original Montreal Protocol agreement (1987), developed countries were required to begin phasing out CFCs in 1993 and achieve a 20% reduction relative to 1986 consumption levels by 1994 and a 50% reduction by 1998. Additionally, developed countries were required to freeze their production and consumption of halons relative to their 1986 levels. After the Montreal Protocol was signed, new data showed worse-than-expected damage to the ozone layer. Because of measures taken under the Montreal Protocol, emissions of ODS are falling and the ozone layer is expected to be fully healed near the middle of the 21st century.
Alternatives to CFCs
HCFCs (hydrochloroflurocarbons)
The original Montreal Protocol, signed in 1987, was the first step in international efforts to protect stratospheric ozone. Under the original Montreal Protocol agreement (1987), developed countries were required to begin phasing out CFCs in 1993 and achieve a 20% reduction relative to 1986 consumption levels by 1994 and a 50% reduction by 1998. Additionally, developed countries were required to freeze their production and consumption of halons relative to their 1986 levels. After the Montreal Protocol was signed, new data showed worse-than-expected damage to the ozone layer. Because of measures taken under the Montreal Protocol, emissions of ODS are falling and the ozone layer is expected to be fully healed near the middle of the 21st century.
Alternatives to CFCs
HCFCs (hydrochloroflurocarbons)
- Positive aspect -break down more quickly in the atmosphere posing less danger to the ozone layer
- Negative aspect -if overused they could cause damage to the ozone layer in the same way as CFC’s.
- Redesigned appliances could use more energy than the original models.
- Positive aspect-do not contain chlorine and are therefore safe for the ozone layer
- Negative aspect -safety questions such as flammability and toxicity still unclear
- Positive aspect – they are relatively cheap and readily available
- Negative aspect -can be flammable and poisonous and some increase ground-level pollution
- Positive aspect -a simple alternative for refrigerators
- Negative aspect -must be handled carefully
- Positive aspect -effective for some cleaning applications
| Ozone biozone worksheet |
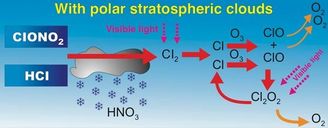
#5 Polar Stratospheric Clouds
Recipe for excess chlorine (Cl), ozone attacking molecules.
- Emissions of Nitrogen Dioxide (NO2)
- CFCs from prior discussed sources
- Ozone
- Methane from anaerobic digestion (CH4)