SOILS
|
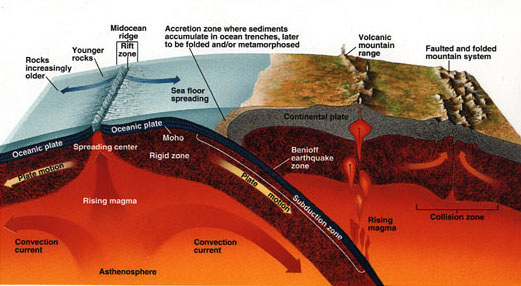
A. The Rock Cycle:
How do we get the minerals content in soil?
Task 2: Types of rocks
How do we get the different types of minerals? Complete the following activity and take notes on the different types of rocks that generate soil. | |||
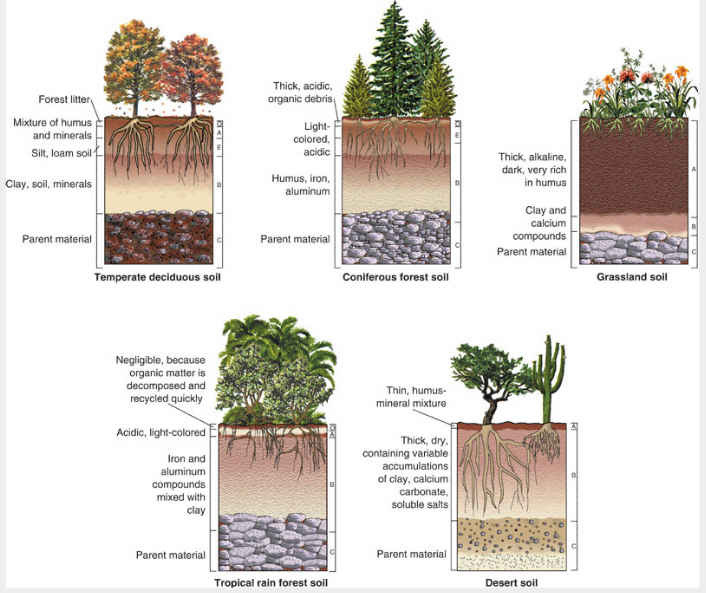
B. Soil Horizons and Profile Development
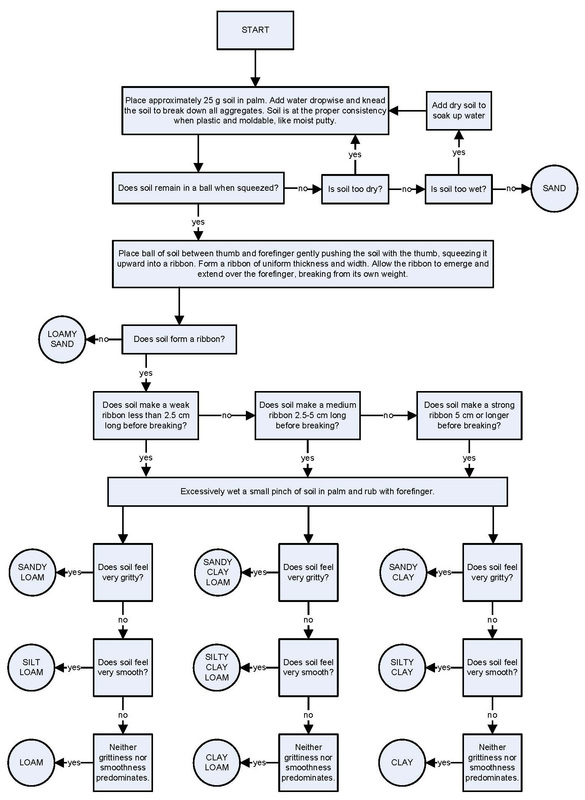
Task 3: What kind of soil do you have in your hand.
Take a soil sample from the teacher or find one yourself. Complete the following palm test yourself.
Take a soil sample from the teacher or find one yourself. Complete the following palm test yourself.
|
|
| ||||||
What kind of soil do we have here?
Justify your answer with the evidence.
Justify your answer with the evidence.
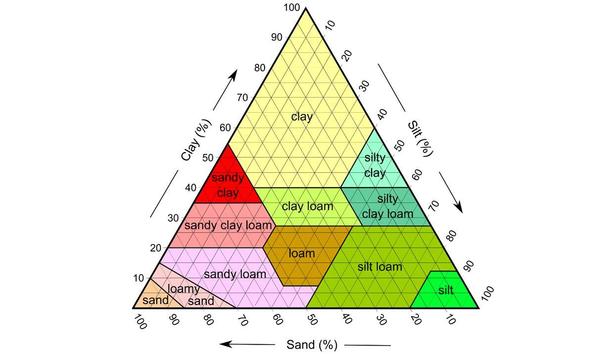
C. Soil Measurement
|
Task 4: What kind of soil do you have?
|
|
|
|
Biotic measurement
Using nature to measure the changes in soil.
Using nature to measure the changes in soil.
|
Hydrangea macrophylla 'Nikko Blue(Biotic indicator species)
Best grown in rich, medium moisture, well-drained soil in part shade. Tolerates full sun only if grown with constant moisture. Soil pH affects flower color (blue color in highly acidic soils and pink color in slightly acidic to alkaline soils). Add aluminum sulfate to soil to make flowers bluer or add lime to soil to make flowers pinker image source: www.homedepot.com |
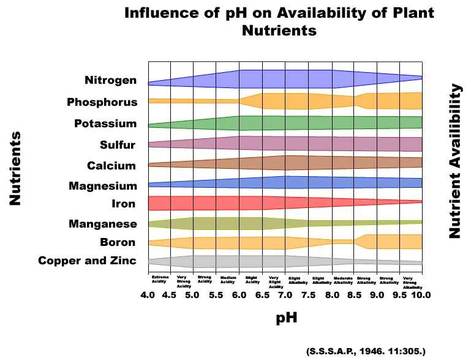 image source: https://onpasture.com/2013/07/15/whats-soil-ph-and-why-do-i-care/
image source: https://onpasture.com/2013/07/15/whats-soil-ph-and-why-do-i-care/
Soil Acidity and it's impacts:
Measuring Soil pH
The most accurate method of determining soil pH is by a pH meter. A second method which is simple and easy but less accurate then using a pH meter, uses litmus papers.
There may be considerable variation in the soil pH from one spot in a field or lawn to another. To determine the average soil pH of a field or lawn it is necessary to collect soil from several locations and combine into one sample.
pH Affects Nutrients, Minerals and Growth
The effect of soil pH is great on the solubility of minerals or nutrients. Fourteen of the seventeen essential plant nutrients are obtained from the soil. Before a nutrient can be used by plants it must be dissolved in the soil solution. Most minerals and nutrients are more soluble or available in acid soils than in neutral or slightly alkaline soils.
But some plants, such as azaleas, rhododendrons, blueberries, white potatoes and conifer trees, tolerate strong acid soils and grow well. Also, some plants do well only in slightly acid to moderately alkaline soils. However, a slightly alkaline (pH 7.4-7.8) or higher pH soil can cause a problem with the availability of iron to pin oak and a few other trees in Central New York causing chlorosis of the leaves which will put the tree under stress leading to tree decline and eventual mortality.
Measuring Soil pH
The most accurate method of determining soil pH is by a pH meter. A second method which is simple and easy but less accurate then using a pH meter, uses litmus papers.
There may be considerable variation in the soil pH from one spot in a field or lawn to another. To determine the average soil pH of a field or lawn it is necessary to collect soil from several locations and combine into one sample.
pH Affects Nutrients, Minerals and Growth
The effect of soil pH is great on the solubility of minerals or nutrients. Fourteen of the seventeen essential plant nutrients are obtained from the soil. Before a nutrient can be used by plants it must be dissolved in the soil solution. Most minerals and nutrients are more soluble or available in acid soils than in neutral or slightly alkaline soils.
- Phosphorus is never readily soluble in the soil but is most available in soil with a pH range centered around 6.5.
- Acidic soils (pH 4.0-5.0) can have high concentrations of soluble aluminum, iron and manganese which may be toxic to the growth of some plants.
- A pH range of approximately 6 to 7 promotes the most ready availability of plant nutrients.
But some plants, such as azaleas, rhododendrons, blueberries, white potatoes and conifer trees, tolerate strong acid soils and grow well. Also, some plants do well only in slightly acid to moderately alkaline soils. However, a slightly alkaline (pH 7.4-7.8) or higher pH soil can cause a problem with the availability of iron to pin oak and a few other trees in Central New York causing chlorosis of the leaves which will put the tree under stress leading to tree decline and eventual mortality.
Changes in Soil pH
Soils tend to become acidic as a result of: (1) rainwater leaching away basic ions (calcium, magnesium, potassium and sodium); (2) carbon dioxide from decomposing organic matter and root respiration dissolving in soil water to form a weak organic acid; (3) formation of strong organic and inorganic acids, such as nitric and sulfuric acid, from decaying organic matter and oxidation of ammonium and sulfur fertilizers. Strongly acid soils are usually the result of the action of these strong organic and inorganic acids.
Lime is usually added to acid soils to increase soil pH. The addition of lime not only replaces hydrogen ions and raises soil pH, thereby eliminating most major problems associated with acid soils but it also provides two nutrients, calcium and magnesium to the soil. Lime also makes phosphorus that is added to the soil more available for plant growth and increases the availability of nitrogen by hastening the decomposition of organic matter. Liming materials are relatively inexpensive, comparatively mild to handle and leave no objectionable residues in the soil.
Some common liming materials are: (1) Calcic limestone which is ground limestone; (2) Dolomitic limestone from ground limestone high in magnesium; and (3) Miscellaneous sources such as wood ashes. The amount of lime to apply to correct a soil acidity problem is affected by a number of factors, including soil pH, texture (amount of sand, silt and clay), structure, and amount of organic matter. In addition to soil variables the crops or plants to be grown influence the amount of lime needed.
In addition to monitoring soil pH the nutrient status of the soil should be examined. To obtain soil sampling instructions and kits along with specific recommendation contact Cornell Cooperative Extension listed in your local phone book under United States Government Offices - Agriculture Department.
source: State University of New York College of Environmental Science and Forestry
Soils tend to become acidic as a result of: (1) rainwater leaching away basic ions (calcium, magnesium, potassium and sodium); (2) carbon dioxide from decomposing organic matter and root respiration dissolving in soil water to form a weak organic acid; (3) formation of strong organic and inorganic acids, such as nitric and sulfuric acid, from decaying organic matter and oxidation of ammonium and sulfur fertilizers. Strongly acid soils are usually the result of the action of these strong organic and inorganic acids.
Lime is usually added to acid soils to increase soil pH. The addition of lime not only replaces hydrogen ions and raises soil pH, thereby eliminating most major problems associated with acid soils but it also provides two nutrients, calcium and magnesium to the soil. Lime also makes phosphorus that is added to the soil more available for plant growth and increases the availability of nitrogen by hastening the decomposition of organic matter. Liming materials are relatively inexpensive, comparatively mild to handle and leave no objectionable residues in the soil.
Some common liming materials are: (1) Calcic limestone which is ground limestone; (2) Dolomitic limestone from ground limestone high in magnesium; and (3) Miscellaneous sources such as wood ashes. The amount of lime to apply to correct a soil acidity problem is affected by a number of factors, including soil pH, texture (amount of sand, silt and clay), structure, and amount of organic matter. In addition to soil variables the crops or plants to be grown influence the amount of lime needed.
In addition to monitoring soil pH the nutrient status of the soil should be examined. To obtain soil sampling instructions and kits along with specific recommendation contact Cornell Cooperative Extension listed in your local phone book under United States Government Offices - Agriculture Department.
source: State University of New York College of Environmental Science and Forestry
Biological components
SOIL FACTORS FOR PLANT HEALTH
What are the necessary ingredients for life? Human beings need water, air, food, and sleep, among other things, to thrive. Just like humans, plants have needs that must be fulfilled in just the correct amounts for them to survive. Water, sunlight, air and nutrients all provide plants with what they need to live and grow.
What are the necessary ingredients for life? Human beings need water, air, food, and sleep, among other things, to thrive. Just like humans, plants have needs that must be fulfilled in just the correct amounts for them to survive. Water, sunlight, air and nutrients all provide plants with what they need to live and grow.