Layers of the atmosphere
|
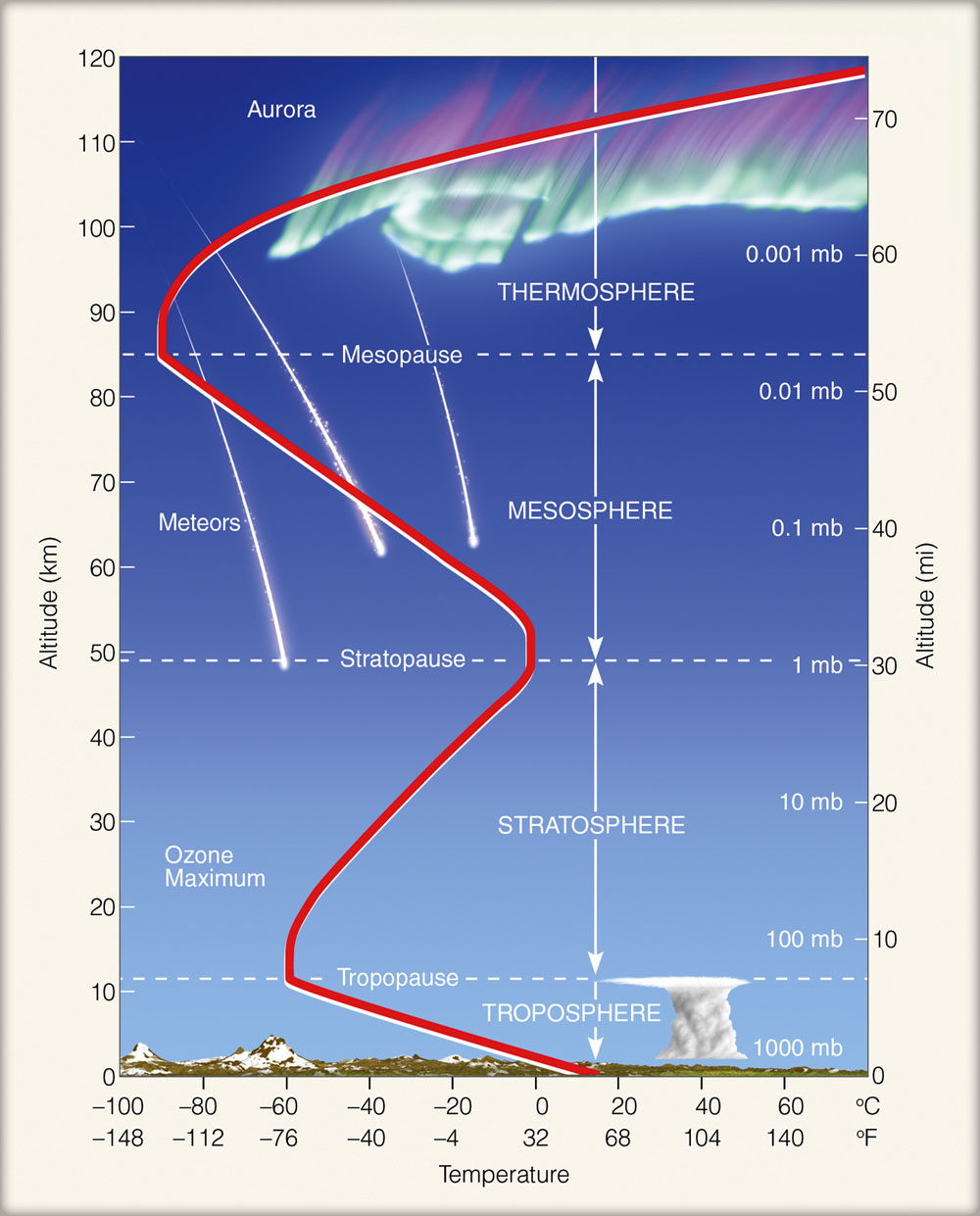
As you move up through the troposphere, it will get colder. This is because the troposphere is heated by the radiation emitted from the ground. The higher you go, that is, the further you get from the source of heat, the cooler it will be. By the time you get to the top of the atmosphere that contains the weather. Above it, the stratosphere is continuously calm and sunny. Within the troposphere both temperature and pressure decrease with height.
As you move up through the stratosphere the temperature will increase because this layer contains a thin band of ozone (the Ozone layer) which along with water vapour absorbs ultraviolet radiation from the sun. The stratosphere is heated from above, so the higher you go, the hotter it will be. The fact that it is warmer than the troposphere means that the stratosphere is relatively stable. The heavier, colder air below it has no tendency to rise and, in effect the stratosphere acts as a lid on the turbulent troposphere. Within the mesosphere, temperature decreases with altitude, as in the troposphere. This is because there are no gases, particles or water vapour to absorb ultraviolet radiation. Within the mesosphere, temperatures drop to their lowest value. Higher up still, into the thermosphere, temperatures increase once more. The upper mesosphere and thermosphere are called the ionosphere, since this layer contains many ions (electrically charged particles), which absorb ultraviolet radiation. Source: Byrne, Kevin. Environmental Science. Cheltenham: Nelson Thornes, 2001. Print.
Task 1: Complete the worksheet on the layers of the atmosphere
|
source: http://geogrify.net/GEO1/Images/FOPG/0314.jpg
| ||||||
|
What's in the air: The Chemical Composition
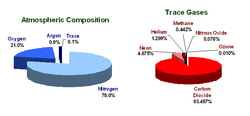
The atmosphere is concentrated at the earth’s surface and rapidly thins as you move upward, blending with space at roughly 100 miles above sea level. The atmosphere is actually very thin compared to the size of the earth, equivalent in thickness to a piece of paper laid over a beach ball. However, it is responsible for keeping our earth habitable and for producing weather. The atmosphere is composed of a mix of several different gases in differing amounts. The permanent gases whose percentages do not change from day to day are nitrogen, oxygen and argon. Nitrogen accounts for 78% of the atmosphere, oxygen 21% and argon 0.9%. Gases like carbon dioxide, nitrous oxides, methane, and ozone are trace gases that account for about a tenth of one percent of the atmosphere. Water vapor is unique in that its concentration varies from 0-4% of the atmosphere depending on where you are and what time of the day it is. In the cold, dry artic regions water vapor usually accounts for less than 1% of the atmosphere, while in humid, tropical regions water vapor can account for almost 4% of the atmosphere. Water vapor content is very important in predicting weather. Greenhouse gases whose percentages vary daily, seasonally, and annually have physical and chemical properties which make them interact with solar radiation and infrared light (heat) given off from the earth to affect the energy balance of the globe. This is why scientists are watching the observed increase in greenhouse gases like carbon dioxide and methane carefully, because even though they are small in amount, they can strongly affect the global energy balance and temperature over time. source: http://climate.ncsu.edu/edu/k12/.AtmComposition |
| ||
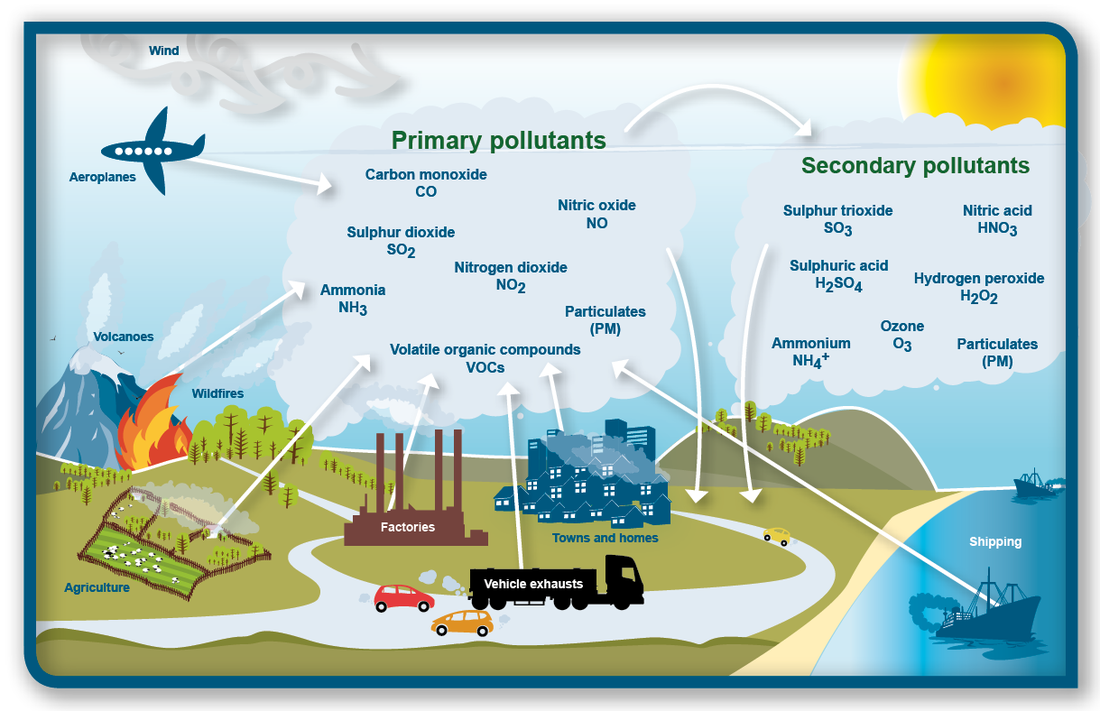
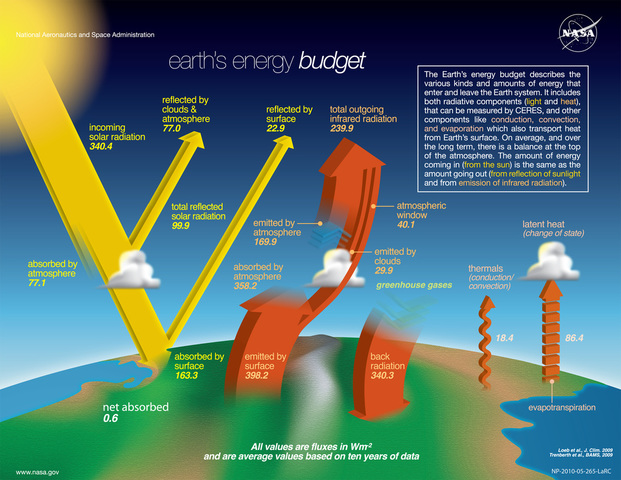
Where does it come from?
source: http://www.nasa.gov/sites/default/files/thumbnails/image/edu_atmosphere_large.jpg
Energy from the sun
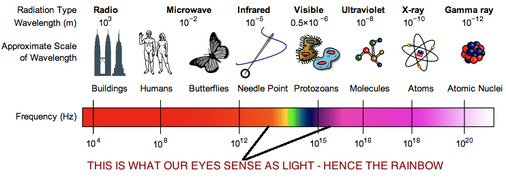
Radiation is the transfer of heat energy by electromagnetic wave motion. The sun emits many forms of electromagnetic radiation in varying quantities. About 43% of the total radiant energy emitted from the sun is in the visible parts of the spectrum. The bulk of the remainder lies in the near-infrared (49%) and ultraviolet section (7%). Less than 1% of solar radiation is emitted as x-rays, gamma waves, and radio waves.
As the temperature increases, the maximum radiation occurs at shorter and shorter wavelengths. The hotter the radiating body, the shorter the wavelength of maximum radiation. For example, a very hot metal rod will emit visible radiation and produce a white glow. On cooling, it will emit more of its energy in longer wavelengths and will glow a reddish color. Eventually no light will be given off, but if you place your hand near the rod, the infrared radiation will be detectable as heat.
The amount of energy absorbed by an object depends upon the following:
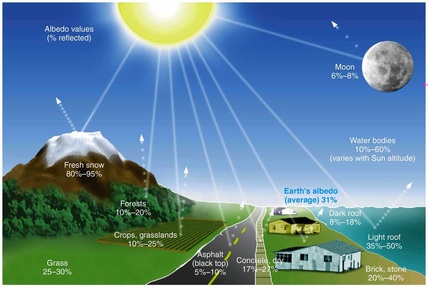
Darker-colored objects absorb more visible radiation, whereas lighter-colored objects reflect more visible radiation. That's why we usually choose light-colored clothing on really hot days.
Every surface on earth absorbs and reflects energy at varying degrees, based on its color and texture.
As the temperature increases, the maximum radiation occurs at shorter and shorter wavelengths. The hotter the radiating body, the shorter the wavelength of maximum radiation. For example, a very hot metal rod will emit visible radiation and produce a white glow. On cooling, it will emit more of its energy in longer wavelengths and will glow a reddish color. Eventually no light will be given off, but if you place your hand near the rod, the infrared radiation will be detectable as heat.
The amount of energy absorbed by an object depends upon the following:
- The object's absorptivity, which, in the visible range of wavelengths, is a function of its color
- The intensity of the radiation striking the object
Darker-colored objects absorb more visible radiation, whereas lighter-colored objects reflect more visible radiation. That's why we usually choose light-colored clothing on really hot days.
Every surface on earth absorbs and reflects energy at varying degrees, based on its color and texture.
Energy is transferred between the earth's surface and the atmosphere via conduction, convection, and radiation. We've already discussed radiation but what about Conduction and convection.
Conduction is the process by which heat energy is transmitted through contact with neighboring molecules.
Some solids, such as metals, are good conductors of heat while others, such as wood, are poor conductors. Air and water are relatively poor conductors. Since air is a poor conductor, most energy transfer by conduction occurs right at the earth's surface. At night, the ground cools and the cold ground conducts heat away from the adjacent air. During the day, solar radiation heats the ground, which heats the air next to it by conduction.
Convection transmits heat by transporting groups of molecules from place to place within a substance. Convection occurs in fluids such as water and air, which move freely. In the atmosphere, convection includes large- and small-scale rising and sinking of air masses and smaller air parcels. These vertical motions effectively distribute heat and moisture throughout the atmospheric column and contribute to cloud and storm development (where rising motion occurs) and dissipation (where sinking motion occurs).
Conduction is the process by which heat energy is transmitted through contact with neighboring molecules.
Some solids, such as metals, are good conductors of heat while others, such as wood, are poor conductors. Air and water are relatively poor conductors. Since air is a poor conductor, most energy transfer by conduction occurs right at the earth's surface. At night, the ground cools and the cold ground conducts heat away from the adjacent air. During the day, solar radiation heats the ground, which heats the air next to it by conduction.
Convection transmits heat by transporting groups of molecules from place to place within a substance. Convection occurs in fluids such as water and air, which move freely. In the atmosphere, convection includes large- and small-scale rising and sinking of air masses and smaller air parcels. These vertical motions effectively distribute heat and moisture throughout the atmospheric column and contribute to cloud and storm development (where rising motion occurs) and dissipation (where sinking motion occurs).
Activity 1: Testing the albedo.